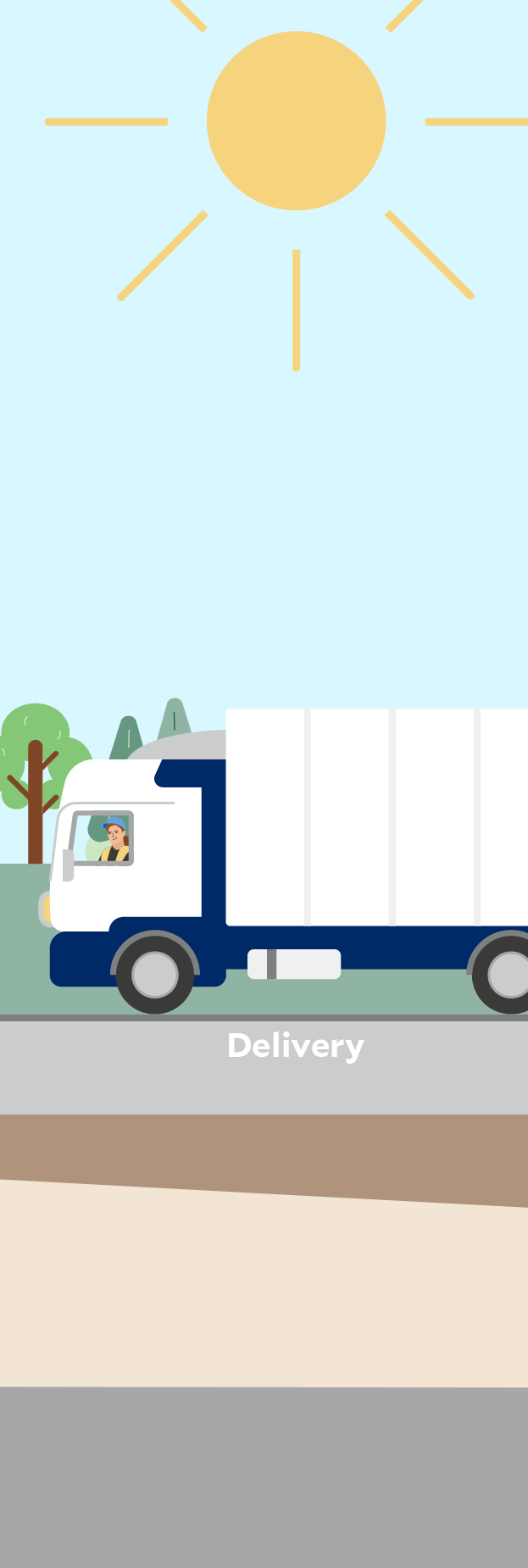
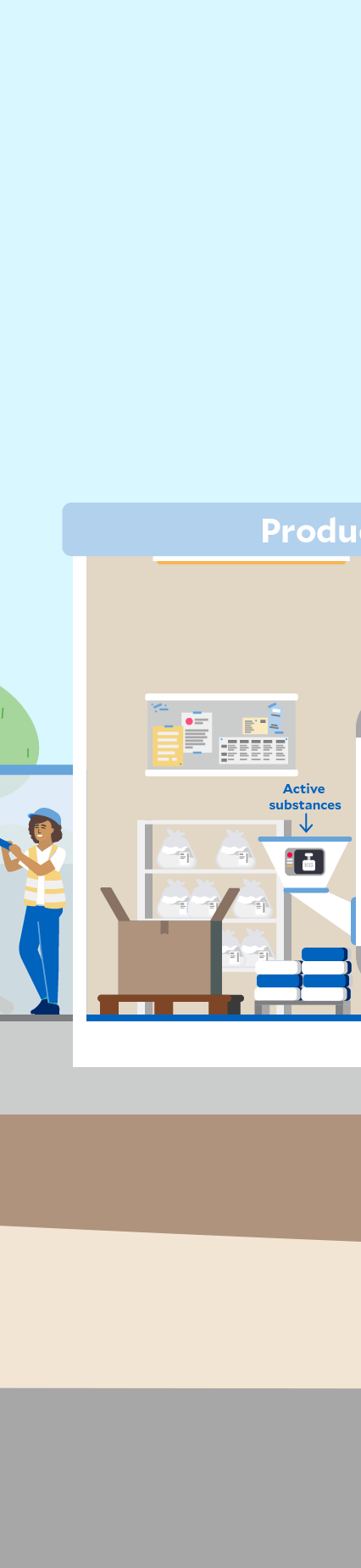
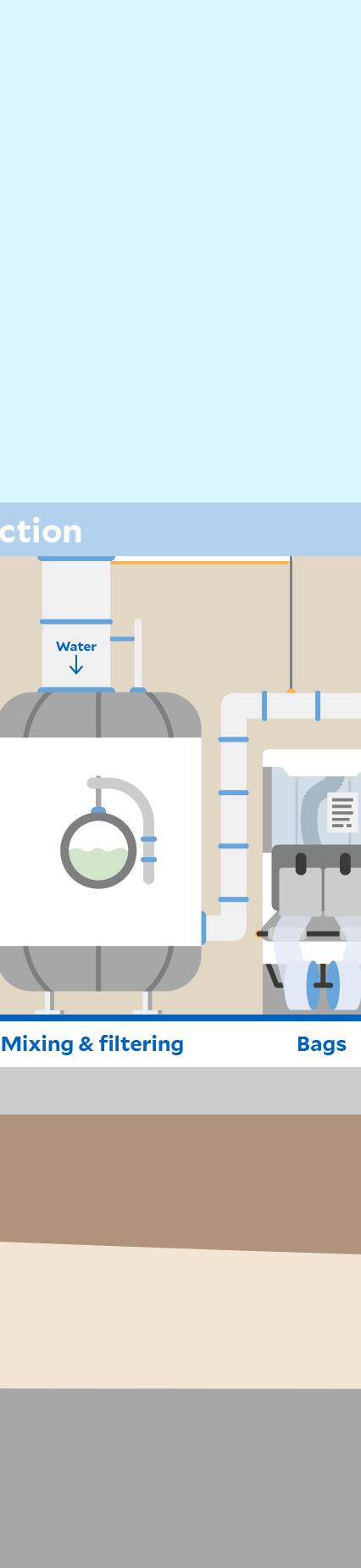
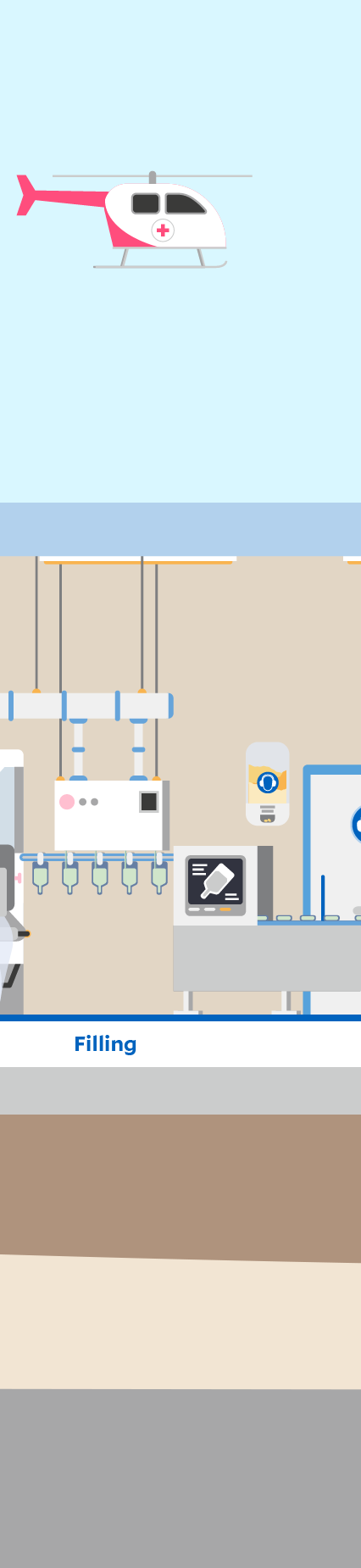
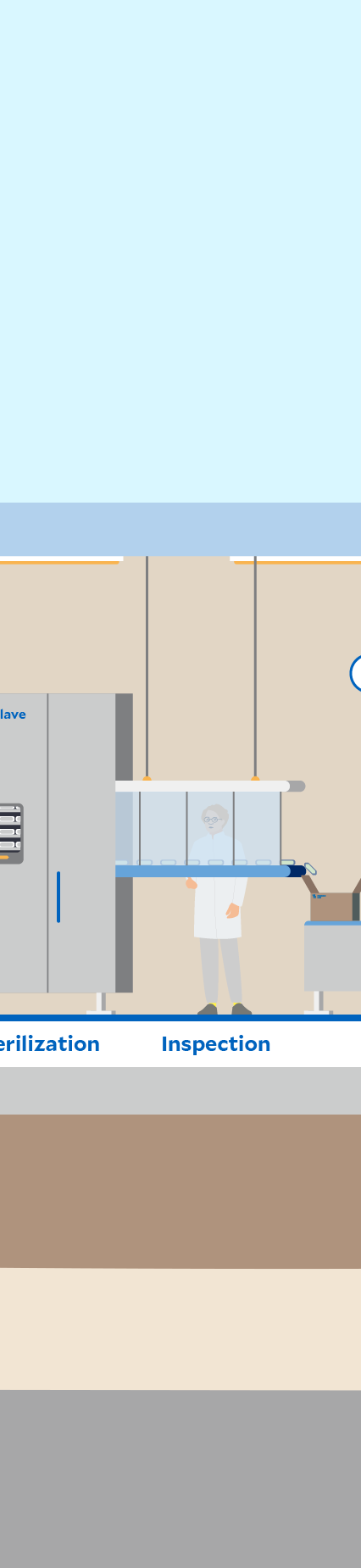
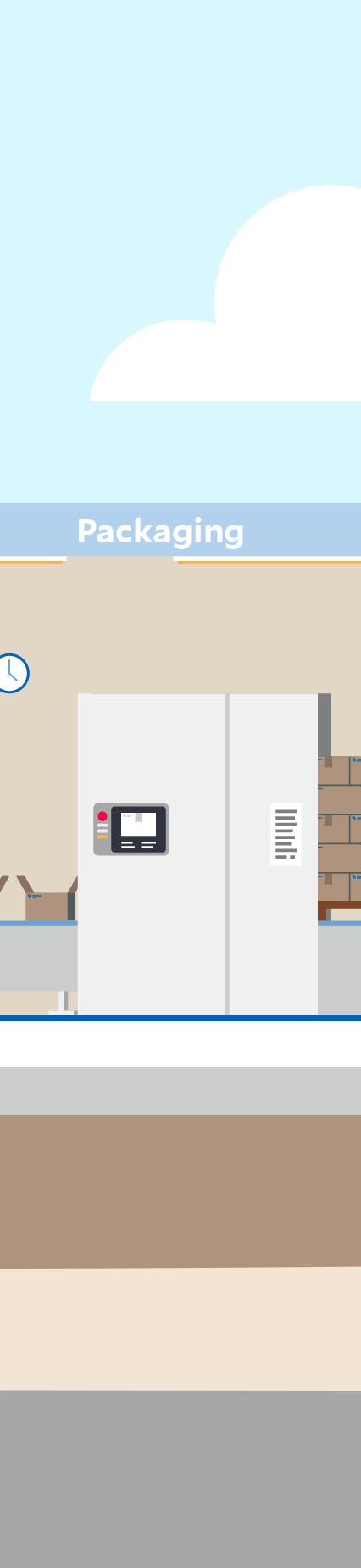
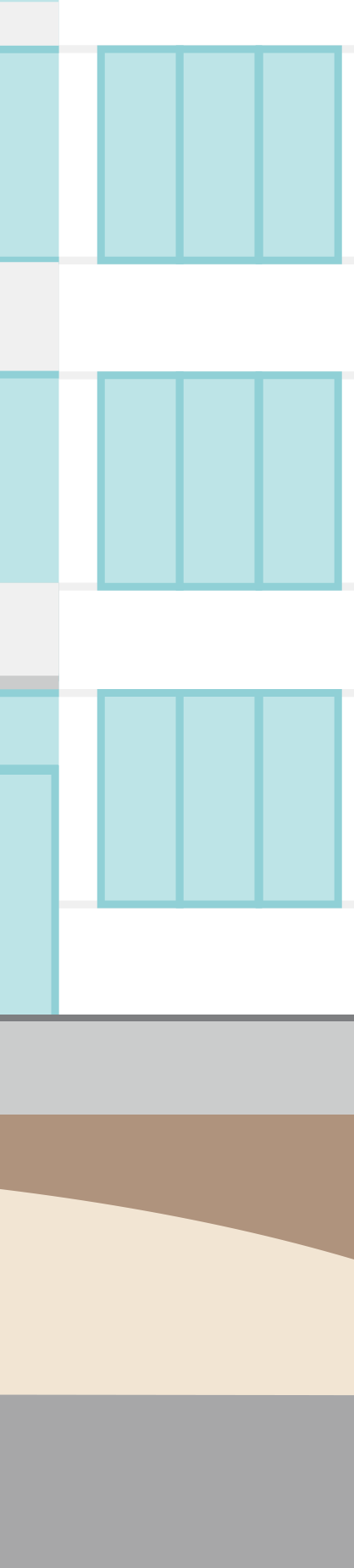
Click through the graphic to find out where sustainability aspects play a role at a Fresenius Kabi production site – here in the manufacturing of infusion solutions as an example.
Emissions in the upstream and downstream value chain – Scope 3

Emissions in the upstream and downstream value chain – Scope 3
We need raw materials and pre-products to manufacture our products, such as nutritional supplements, infusion solutions, infusion bags and drugs. The finished products are in turn supplied via distributors to pharmacies, hospitals and health centers and ultimately to patients for use. All these steps generate indirect greenhouse gas emissions, known as Scope 3 emissions.
Further informationHuman rights

Human rights
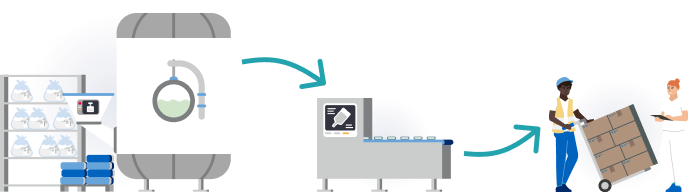
Our commitment to respecting human rights extends beyond our own corporate activities and our core business. For example, we source raw materials and intermediate products for production from other Fresenius Kabi sites as well as from external suppliers. In line with our human rights program, we take human rights concerns into account when selecting and working with our suppliers and business partners.1
Further information1Detailed information on the measures of our human rights program can be found in our audited Sustainability Statement 2025.
Product development
Product development
The further or new development of products and processes is an integral part of our strategy. Developments take place at various levels. In the area of biosimilars, we are dedicated to the development of cost-effective drugs based on reference products.
Further informationWater and wastewater
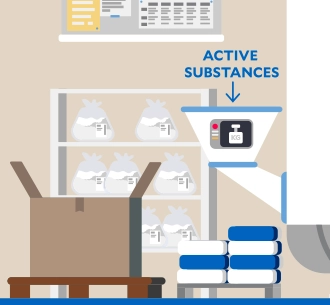
Water and wastewater
Water is a valuable resource. To ensure the quality of our products, we depend on the availability of water. That is why we have set ourselves the target of reducing process water. We aim to reduce absolute process water withdrawal at production sites in areas with water stress by 20% by 2030 (base year: 2023; baseline value: 3,518,096 m3).1 At the same time, we are responsible for the proper handling of wastewater from production.
Further information Highlight Story: Resistant pathogens: Prevention in hospitals when manufacturing antibiotics1We report in detail on our reduction target, the measures planned to achieve it, and current progress in our audited Sustainability Statement 2025.
Automation and digitalization
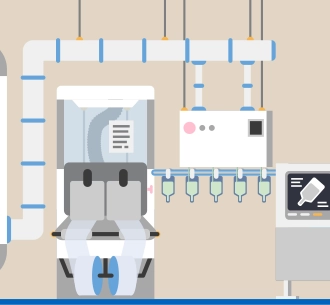
Automation and digitalization
Automation and digitalization can support process stability and product quality. In production, for example, sections such as filling, sterilization, and packaging are completely automated.
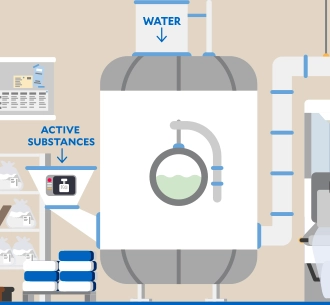
Energy and climate
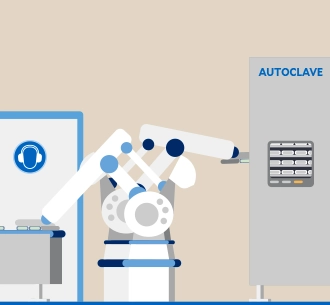
Energy and climate
High-energy processes are used at our production sites, such as the sterilization of infusion solutions in an autoclave. In order to reduce the greenhouse gas emissions released by these processes as far as possible, we are gradually driving forward energy efficiency measures as well as the use of renewable energy and the expansion of photovoltaic systems. These are levers for our group-wide climate targets.1
Further information1Detailed information on our climate targets and our climate transition plan can be found in our audited Sustainability Statement 2025.
Quality of the products
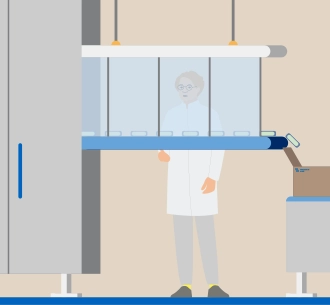
Quality of the products
Patient safety and therefore product quality are our top priority. Structured processes, training and comprehensive quality management are fundamental to this. Regular checks are carried out at our sites to ensure that all relevant standards and regulations are complied with.
Further informationResource management
Resource management
We source various raw materials and intermediates for the manufacture of our healthcare products. Waste is mainly generated as by-products of production processes or as packaging material in the downstream value chain.
Further informationOccupational safety and working conditions
Occupational safety and working conditions
We want to offer our employees a safe and healthy working environment. We support them in their personal development with appropriate programs through a culture of learning.
Further informationAccess to healthcare
Access to healthcare
It is our task to develop products that help to promote medical progress in acute and follow-up care and improve the quality of life of patients. At the same time, we want our products to enable more and more people around the world to have access to high-quality and modern therapies.
Further information Highlight Story: How Fresenius Kabi supports access to essential therapies in everyday care Highlight Story: When every day counts: EASYGEN is set to make cell therapy more accessibleResponsible partnerships

Responsible partnerships
We act in accordance with our Code of Conduct and the Fresenius Principles. We expect our business partners to commit to our ethical principles in accordance with the Business Partner Code of Conduct. At the same time, we promote cooperation with NGOs, public authorities, and other companies to drive innovation and solutions along the value chain.
Further information